For some time now, Motorola, NEC, Toshiba and other companies have been working on how to provide more power to mobile handsets and laptops for longer periods of time. Conventional battery technology has just about been pushed to its limits – after all, if a chemical battery could somehow store a lot of power, it would probably be so unstable that at full charge it would be always be on the verge of exploding.
Most attempts at extending the operating time of mobile devices have centered on fuel cells, which convert hydrogen and oxygen into water, generating electricity in the process. Prototype fuel cells don’t actually use tanks of hydrogen. Instead small battery-sized cartridges of methanol are used. In such Direct-Methanol Fuel Cells (DMFC), methanol is oxidized on a catalyst layer to form carbon dioxide. Water is consumed at the anode and is produced at the cathode. Positive ions (H+) are transported across the proton exchange membrane to the cathode where they react with oxygen to produce water. Electrons are transported through an external circuit from anode to cathode, providing power to connected devices. Methanol is easier to store than hydrogen, which requires high pressures or low, cryogenic temperatures. Methanol is a liquid at room temperature, and its energy density (the amount of energy contained in a given volume) is an order of magnitude greater than even highly compressed hydrogen.
Companies such as Entegris ( News - Alert) (www.entegris.com) specialize in polymer material science and have expertise in providing advanced fuel cell materials, components, subassemblies and value-added services to fuel cell developers.
The only downside is that a fuel-cell powered phone would produce some carbon dioxide and water. Carrying around a cell phone capable of wetting your clothes sounds a bit disturbing, but there are various ways of capturing or causing the water to evaporate without creating a mess. Fuel cells would be quite “green” and environmentally friendly (if also a bit damp).
If you think a fuel cell telephone would be a bit too wet, you should have seen the “water-jet” transmitter (telephone) developed by Dr. Chichester A. Bell, cousin of Alexander Graham Bell. He based his invention of research which can be found in a paper he read before the British Royal Society in 1886. The phone is quite bizarre. By adding a bit of sulphuric acid to water (thus making it electrically conductive) Bell discovered that the electrical resistance of a jet of conducting fluid by mechanical (in this case, sound) vibrations.
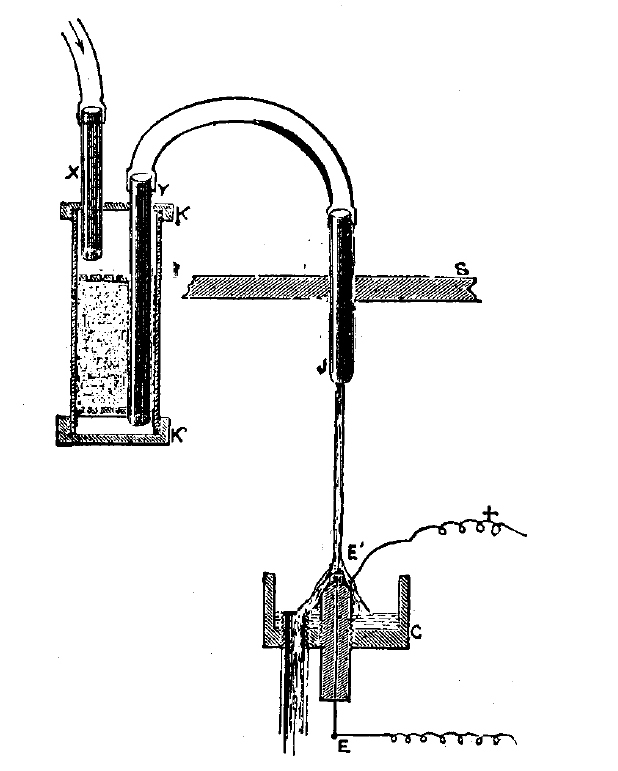
A stream of water falls from a tube onto a platinum contact. When the water strikes the contact, the water sprays out horizontally in all directions and then strikes a platinum ring surrounding the contact. By passing an electrical current through the stray to the ring, Bell discovered that a tiny deflection of the jet caused a great variation in the current flowing through this “water circuit” (see the schematic). By deflecting the jet with sound waves, he could create electrical variations matching the sound and thus build a “Water-Jet Telephone” on this principle (see illustration).
The Water-Jet phone was about three feet high and mounted on a wall. A tank at the top of the phone held a water supply. At its base, a second tank stored the used water. Water could be recycled back up to the top tank by a lever-operated pump. A single movement of the lever filled the reservoir with enough water for a seven minute phone call. If the call took longer, the user could just pump again during the call.
Fuel cells don’t sound so bad now, do they?
Richard Grigonis is Executive Editor of TMC (News - Alert)’s IP Communications Group. To read more of Richard’s articles, please visit his columnist page.
Edited by Stefania Viscusi
|