TMCnet News
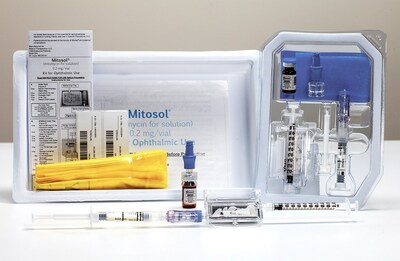
Mobius Therapeutics™ Customers to Advocate Use of Mitosol®First use of social media platform by Mobius™ supports value proposition to end users. ST. LOUIS, May 10, 2022 /PRNewswire/ -- Mobius Therapeutics™, LLC, a St. Louis-based perioperative ophthalmic pharmaceutical company, will initiate a social media presence featuring interviews and live streaming of customers to support the value proposition of its flagship product, Mitosol®. With Mitosol®, everyone wins: the patient, the provider, and the American healthcare system. "Mitosol® customers have the option to purchase compounded copies of our approved formulation from pharmacies and outsourcing facilities," said Ed Timm, CEO of Mobius Therapeutics. "Their loyalty to Mitosol® reflects its inherent value to patients and caregivers. Providers need to hear real world views from their peers, not just company authored advocacy." Mitosol® is the only FDA approved formulation of mitomycin-c bearing an ophthalmic indication. With room temperature storage and shelf life of up to 24 months, Mitosol® offers unique flexibility to providers, as its "shelf-ready" status permits on demand utility. "Real-world voices are indicative of real-world value," continued Timm. "We must listen to these voices, as they translate marketing into common vernacular. They tell us that with Mitosol®, everyone wins: the patient, the provider, and the American healthcare system." About Mobius Therapeutics, LLC: CONTACT:
SOURCE Mobius Therapeutics, LLC 
|